A photonic picocavity in action
Light emission from emitters is a valuable piece of information in a variety of sensing and detection techniques, capable of labeling physical and biological processes which occur in the proximity of the emitter. Furthermore, quantum technologies are currently exploiting the statistics of single-emitters light emission aiming at turning particular emitters into fundamental units to sustain qubits-based quantum technology. In this context, molecular quantum emitters show the possibility of coupling with cavity photons due to their strong dipole moments, particularly when located in the proximity of nanoscale resonators such as in plasmonic nanocavities where ultraconfined effective cavity mode volumes can be achieved. This coupling boosts the interaction between the emitting molecular excitons and cavity plasmons, producing an enhancement or quenching of light emission, and even polaritonic hybridization of states.
Molecular photoluminescence is a process driven by light which creates an electronic transition in a molecule, further relaxing and emitting a new photon of lower energy associated with the electronic transition from the unoccupied molecular orbital (LUMO) to the highest occupied molecular orbital (HOMO) of the molecule. In a sophistication of the nanoscale resonator concept, a research team from the University of Science and Technology of China (Hefei, China), in collaboration with researchers from the Max Planck Institute for the Science of Light (Erlangen, Germany) and from the Center for Materials Physics (San Sebastian, Spain) have engineered a special type of atomic-scale cavity in a tunneling plasmonic junction, so-named “picocavity”, constituted by an atomic protrusion at the apex of a metallic tip, which allowed for exploring the properties of molecular emission and map them spatially with unprecedented resolution.
By depositing a single zinc phthalocyanine (ZnPc) on a silver surface conveniently detached from the metal by three layers of NaCl in order to avoid non-radiative quenching, together with a precise positioning of the tunneling tip with respect to the molecule, researchers scanned the controllable picocavity over the molecule, mapping the light emitted with submolecular resolution, as observed in the figure.
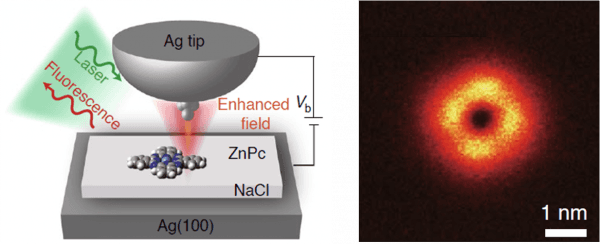
Figure. (Left) Schematics of the single molecule photoluminescence measurement setup. A picocavity formed by a silver tip with an atomic protrusion is located on top of a silver surface with a ZnPc molecule on it, separated by a NaCl layer. The tunneling picocavity scans the molecule and provides a tip position-dependent fluorescence map with atomic-scale resolution. (Right) Single ZnPc photoluminescence map with submolecular resolution obtained with the setup on the left.
This special picocavity produces an effective enhancement of the light emission from the ZnPc molecule (Purcell effect) which depends on its actual position over the molecule within Ångstrom precision. As observed in the figure, the picocavity is able to map the light emission from a single ZnPc, resolving the four-fold symmetry of the molecule.
The research team analyzed the dependence of the light emitted by the molecule for different positions of the tunneling tip, and derived the intensity and decay of the near field achieved at the picocavity, confirming the ultraconfined nature of this extreme photonic resonator. Compared to previous studies on single-molecule light emission from tunneling junctions (electroluminescence), in this work the excitation of the excitonic emission is purely optical (bias voltage below the emission threshold), thus these results fulfill the long-standing dream of the scanning near-field microscopy (SNOM) community which has aimed for decades at resolving the internal structure of a single molecule by the action of an extremely confined optical near field, as the one achieved in the picocavity.