Impact of temperature and architecture on the interfacial structure of microgels: A neutron reflectometry and computer simulations study
This study provides an in-depth microscopic description of how different microgel architectures affect their arrangements at interfaces, and will be the foundation for a better understanding of their phase behavior and assembly. Both aspects are crucial to preparing smart emulsions that can be broken on demand.
Soft microgels—cross-linked polymer networks swollen in a good solvent—reveal peculiar properties that are different from those of other colloidal systems such as
hard nanoparticles, polymers, and surfactants. Particularly, microgels based on poly-N-isopropylacrylamide (pNIPAM) have a high interfacial activity and at the same time maintain their thermo-responsiveness once adsorbed to air, liquid-, or solid interfaces.
The structural characterization of pNIPAM microgels at interfaces is fundamental to understand both their 2D phase behavior and their role as stabilizers that enable emulsions to be broken on demand as a function of external stimuli such as temperature and pH. However, this characterization is usually limited by available experimental techniques, which do not allow a direct investigation at interfaces. To overcome this difficulty, the authors employ neutron reflectometry (NR), which allows them to probe the structure and responsiveness of the microgels in-situ at the air-water interface. Bochenek et al. investigate two types of microgels with different cross-link density, thus having different softness and deformability, both below and above their volume phase transition temperature, by combining NR experiments with computer simulations of in silico synthesized microgels. The advantage of NR is that it allows to probe the structure of a statistically significant ensemble of microgels in situ at the interface. Using NR, we can directly measure the protrusion of the microgels in the air and estimate how it changes with temperature. Microscopy-based techniques, such as transmission X-ray microscopy (TXM) or cryo-SEM, are usually limited by the small number of observed particles, the size of the particles, an observation direction perpendicular to the interface, and complicated sample preparation.
In this contribution, they found that temperature only affects the portion of microgels in water, while the strongest effect of the microgels softness is observed in their ability to protrude into the air. In particular, standard microgels have an apparent contact angle of few degrees, while ultra-low cross-linked microgels form a flat polymeric layer with zero contact angle.
The differences highlighted in the structure may be relevant for a more comprehensive understanding of microgels’ effective interactions, paving the way for a better description of their 2D assembly and for a clever design of their applications such as emulsion stabilizers.
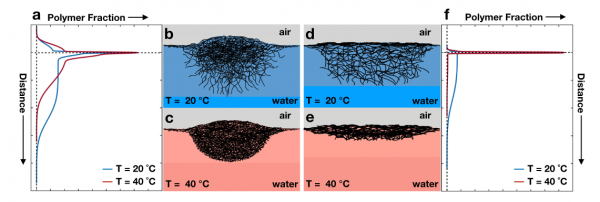
Figure: Sketch of the adsorbed microgels. a The vertical profiles of standard microgels and f the vertical profiles of ULC microgels below and above the VPTT. Their corresponding shapes are outlined in b–e. The shapes are based on the combination of the team’s polymer fraction profiles, simulations and AFM measurements at the liquid-solid interface from the literature.