Single-chain nanoparticles: opportunities provided by internal and external confinement
Single-Chain Nanoparticles (SCNPs) result from the folding individual synthetic polymer chains by means of multiple intra-chain (reversible or irreversible) bonding interactions. The size of these soft nano-objects can be easily tuned between 3 and 30 nm. Only a rough analogy exists between the process of SCNP formation and the precise, specific folding of a polypeptide chain to its native, functional state (e.g., enzymes). Most SCNPs result in a typical morphology in solution more akin to those displayed by Intrinsically Disordered Proteins (IDPs).
Strategies to induce globule formation in SCNPs were also investigated, most of them based on tuning the hydrophobic/hydrophilic balance of the SCNP precursor. The conformational degrees of freedom in SCNPs are severely restricted, giving rise to notorious local domain formation and, hence, to interesting topological self-confinement effects.
Self-confinement in SCNPs has already been exploited for catalysis, encapsulation, transport and delivery of therapeutic cargos, sensing, targeting and bioimaging applications, as well as a variety of other advanced applications. Further progress is expected in these fields accompanied by the development of new methods of rigorous sequence control and precise placement of single functional monomer units at multiple positions along the polymer backbone. This development will presumably lead to precise control of the SCNP topology and, hence, to highly accurate structure–property relationships.
External confinement – as imposed under different geometrical constraints as well as in crowded solutions, in the melt state and on surfaces – is an extra parameter to tune the size, shape, dynamics and, hence, potential functionality/foreseen applications of SCNPs. The unifying vision that results by combining recent experiments, simulations and theory suggests that, in addition to the global size and shape, external confinement also affects the local internal structure of SCNPs and their dynamics, which are critical e.g. for catalysis and drug delivery applications. This recent knowledge opens up new avenues to tune the properties and functionality of SCNPs (see Figure).
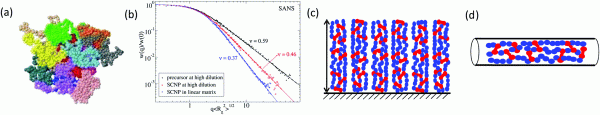
Figure: Illustration of external confinement imposed on SCNPs: (a) Crumpled globule morphology of sparse SCNPs (drawn in different colours) in a concentrated solution as revealed by MD simulations. (b) Change in conformation of SCNPs on passing from dilute solution (sparse open conformation, scaling exponent ν = 0.46) to all-polymer nanocomposites (crumpled globule conformation, scaling exponent ν = 0.37) as determined by SANS experiments. (c) Schematic illustration of arrays of SCNPs on surfaces. (d) Change in conformation of SCNPs in nanopores.
Hence, innovative purification techniques of SCNPs can be envisioned based on their expected behavior during ultrafiltration through nanopores under an elongational flow field. The compact, crumpled globule conformation that sparse SCNPs, adopt under crowding in the melt and in all-polymer nanocomposites ,often induces significant viscosity reduction effects that could be beneficial for the development of improved all-polymer nanocomposites. Smart responsive surfaces could be foreseen based on a dense array of SCNPs attached to a surface loaded with active substances to be delivered through stimuli-mediated rupture of intra-chain cross-links. Moreover, new topological nanostructures could result by increasing the complexity of the polymer precursors used for SCNP synthesis, their hydrophobic/hydrophilic balance, self-assembly properties, and so on.